- Luật
- Hỏi đáp
- Văn bản pháp luật
- Luật Giao Thông Đường Bộ
- Luật Hôn Nhân gia đình
- Luật Hành Chính,khiếu nại tố cáo
- Luật xây dựng
- Luật đất đai,bất động sản
- Luật lao động
- Luật kinh doanh đầu tư
- Luật thương mại
- Luật thuế
- Luật thi hành án
- Luật tố tụng dân sự
- Luật dân sự
- Luật thừa kế
- Luật hình sự
- Văn bản toà án Nghị quyết,án lệ
- Luật chứng khoán
- Video
- NGHIÊN CỨU PHÁP LUẬT
- ĐẦU TƯ CHỨNG KHOÁN
- BIẾN ĐỔI KHÍ HẬU
- Bình luận khoa học hình sự
- Dịch vụ pháp lý
- Tin tức và sự kiện
- Thư giãn

TIN TỨC
fanpage
Thống kê truy cập
- Online: 223
- Hôm nay: 198
- Tháng: 1621
- Tổng truy cập: 5245625
mRNA cancer therapies planned for 2030
BioNTech has announced a strategic partnership with the UK government to provide up to 10,000 patients with personalised mRNA cancer immunotherapies by 2030.
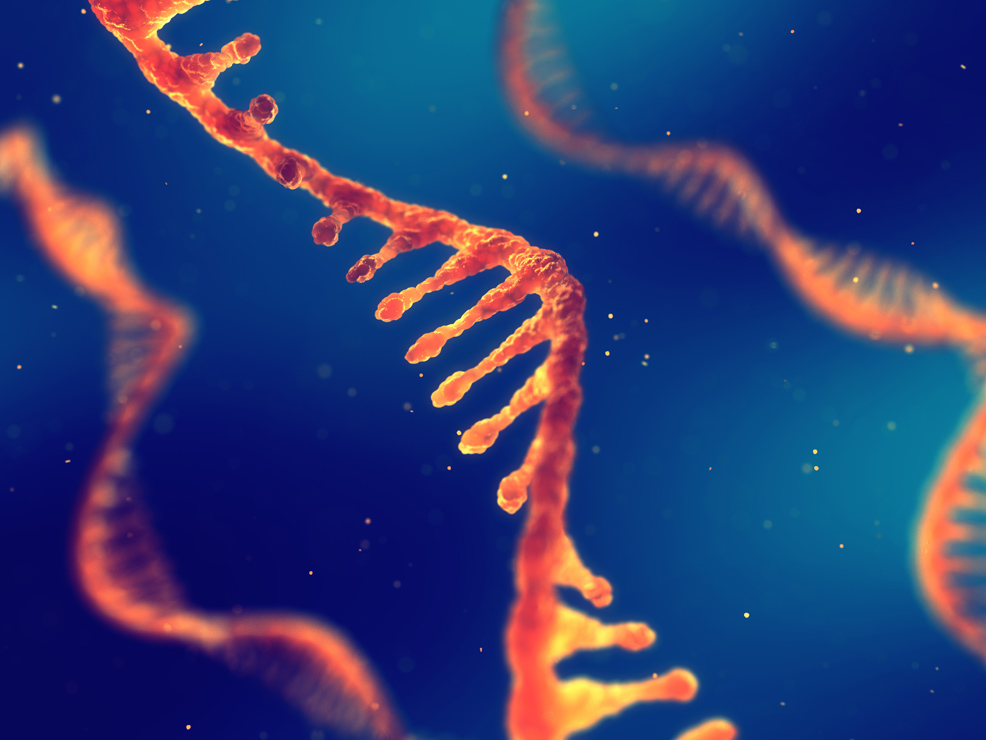
BioNTech is famous for having partnered with Pfizer to develop a COVID-19 vaccine, based on messenger RNA (mRNA). More recently, the company has been further developing this technology to investigate its potential in treating other diseases, such as cancer. One study, which involved the injection of mRNA into colon and melanoma tumours in mice, halted tumour growth and caused a complete regression of cancer in 85% of the animals. A phase 1 trial in 231 humans is currently underway and expected to conclude in 2024.
This week, BioNTech signed an agreement with the UK government to accelerate patient clinical trials of mRNA immunotherapies. The aim is to significantly improve cancer outcomes by providing new personalised therapies, as well as treating a much greater number of people – in this case, up to 10,000 – either in clinical trials or as authorised treatments by 2030.
To achieve this, BioNTech plans to utilise the UK's clinical trial network, world-leading genomics, and health data assets. The next steps of the collaboration will be the selection of candidates, trial sites, and a development plan to ensure the first cancer patient is ready to be enrolled in the second half of 2023.
Traditional chemotherapy involves a widespread attack on many different cells, as well as the cancer, which can lead to significant side effects. By contrast, treatments with mRNA are potentially safer, and personalised for the individual. A drug can provide the body's immune system with pieces of genetic code from a specific cancer, so it "learns" to attack only the tumour, making it far more targeted and precise.
"The UK successfully delivered COVID-19 vaccines so quickly because the National Health Service, academia, the regulator and the private sector worked together in an exemplary way," said Prof. Ugur Sahin, M.D., CEO and Co-Founder of BioNTech. "This agreement is a result of the lessons learnt from the COVID-19 pandemic as we all experience that drug development can be accelerated without cutting corners if everyone works seamlessly together towards the same goal. Today's agreement shows that we are committed to do the same for cancer patients.
"Our goal is to accelerate the development of immunotherapies and vaccines using technologies we have been researching for over 20 years. The collaboration will cover various cancer types and infectious diseases, affecting collectively hundreds of millions of people worldwide. If successful, this has the potential to improve outcomes for patients and provide early access to our suite of cancer immunotherapies, as well as to innovative vaccines against infectious diseases – both in the UK and worldwide."
BioNTech is creating a new research and development hub in Cambridge, one of the UK's most high-tech cities, with an expected capacity of more than 70 highly skilled scientists. R&D will commence by the end of Q1 2023. In addition, the company will strengthen its UK presence by establishing a new regional headquarters in London, to accommodate employees in global and regional supporting functions.
"mRNA vaccines are one of the most exciting research developments to come out of the pandemic, and there are strong hints that they could become powerful treatment options for cancer," said Dr Iain Foulkes, spokesperson for Cancer Research UK. "Getting there will require lots more research."
By FutureTimeLine
Các bài viết khác
- Vài nét Dự báo thời đại phục hưng và khai sáng của loài người sau đại dịch Corona-2019.7-2021(khởi đầu từ tháng 9 năm giáp thìn 2024) (25.06.2021)
- Từ sự kiện Tổng biên tập báo TIME Greta Thunberg là Nhân vật của năm 2019 đến báo cáo Biến đổi khí hậu Phúc trình của IPCC báo động đỏ cho nhân loại 82021 (15.01.2020)
- Loại bỏ CO2 cần tăng 1.300 lần vào năm 2050 CO2 removal needs 1,300x increase by 2050 (25.01.2023)
- Mộng du trên đỉnh núi Đại thảm họa thế kỷ 21 Sleepwalking on Megathreat Mountain (25.01.2023)
- Giới tinh hoa thế giới tại Davos nên thức tỉnh trước Đại thảm họa nhân loại thế kỷ 21 Davos elites need to wake up to megathreats the world is facing (25.01.2023)


















































 Yahoo:
Yahoo: 